Induced Native Phage Therapy (INPT) and related Inducen® formulations are intended to
function as an important key component of a broader, integrative, non-cytotoxic therapeutic
strategy. They are not proposed as standalone cancer treatment, but as adjunctive interventions
designed to address contributory microbial drivers of oncogenesis while preserving commensal
microbiome integrity.
List of 10 Bacterial Infections That Can Instigate Cancer
Below is a reproduced and updated list of various bacterial infections known or suspected to
instigate cancer, based on our previous discussions and peer-reviewed research from the web
search results. The list focuses on key bacteria, the associated cancer types, and the mechanisms
by which they initiate or promote carcinogenesis. These mechanisms often involve chronic
inflammation, DNA damage, immune evasion, and oncogenic signaling pathways. Note that
while some associations are causative (e.g., Helicobacter pylori as a Class I carcinogen by
WHO), others are correlative and require further validation.
1. Helicobacter pylori
- Associated Cancer Types: Gastric (stomach) cancer, gastric MALT lymphoma.
- Initiation Mechanism: Induces chronic inflammation through CagA oncoprotein,
which activates NF-κB and Wnt/β-catenin signaling, leading to mucosal damage,
oxidative stress, DNA methylation, p53 mutations, and oncogenic transformation.
Also downregulates DNA repair pathways and produces reactive oxygen species
(ROS) for genomic instability.
2. Salmonella typhi
- Associated Cancer Types: Gallbladder cancer.
- Initiation Mechanism: Produces cytolethal distending toxin B (CdtB) and biliary
deoxycholate metabolites, causing p53 gene mutations, protein kinase activation,
and upregulation of the PI3K pathway, leading to chronic inflammation, DNA
damage, and cell proliferation.
3. Streptococcus bovis / gallolyticus - Associated Cancer Types: Colorectal cancer, endometrial cancer.
- Initiation Mechanism: Produces pro-inflammatory metabolites like lactate,
upregulates COX-2, and induces cytokine secretion (e.g., IL-8, TNF-α),
promoting angiogenesis, chronic inflammation, and NF-κB activation, which
drives cell proliferation and tumor initiation (, , , , ).
4. Chlamydia pneumoniae
- Associated Cancer Types: Lung cancer.
- Initiation Mechanism: Increases cytokine secretion (IL-8, IL-10, TNF-α),
overexpresses miRNA-328, activates lung-resident T-cells, synthesizes MyD88-
dependent IL-1β and IL-23, and produces ROS, leading to chronic inflammation,
oxidative stress, and genomic instability.
5. Escherichia coli (pks+ strains)
- Associated Cancer Types: Colorectal cancer.
- Initiation Mechanism: Produces colibactin (clbB toxin), causing DNA double-
strand breaks, genomic instability, and NF-κB-mediated inflammation; also
induces catenin stimulation of IL-17R, NF-κB, and STAT3 signals, promoting
cell proliferation and tumorigenesis (, , , , ).
6. Bacteroides fragilis (enterotoxigenic strains)
- Associated Cancer Types: Colorectal cancer.
- Initiation Mechanism: Produces Bacteroides fragilis toxin (BFT), which
activates NF-κB (increasing IL-17A and TNF-α), STAT3 signaling, and EMT;
induces DNA damage and catenin stimulation of IL-17R, NF-κB, and STAT3,
leading to chronic inflammation and tumor progression (, , , , ).
7. Porphyromonas gingivalis
- Associated Cancer Types: Oral cancer, esophageal cancer, pancreatic cancer.
- Initiation Mechanism: Secretes gingipains that degrade extracellular matrix,
suppress apoptosis, induce IL-8 for angiogenesis, and promote chronic
inflammation through NF-κB activation, leading to tissue invasion and oncogenic
transformation.
8. Chlamydia trachomatis
- Associated Cancer Types: Cervical cancer (often with HPV co-infection).
- Initiation Mechanism: Causes chronic inflammation and oxidative stress,
weakening the immune system and facilitating HPV persistence, leading to
genomic instability and oncogenic changes in cervical cells.
9. Fusobacterium nucleatum
- Associated Cancer Types: Colorectal cancer, pancreatic cancer, breast cancer,
lung cancer, gastric cancer. - Initiation Mechanism: Produces FadA and Fap2 virulence factors that activate
Wnt/β-catenin signaling, induce chronic inflammation (IL-6, TNF-α, IL-8),
promote EMT, suppress immune responses (e.g., TIGIT binding on NK-cells),
and cause DNA damage, leading to cell proliferation and metastasis.
10. Clostridium septicum
- Associated Cancer Types: Colorectal cancer.
- Initiation Mechanism: Induces gas gangrene-like necrosis and inflammation,
promoting NF-κB and STAT3 signaling, which drives cell proliferation and tumor
initiation in the gastrointestinal tract (, , ).
This list is not exhaustive but highlights some of the most well-established associations.
Bacterial infections contribute to ~15–20% of global cancers, primarily through chronic
inflammation and DNA damage. For INPT targeting, focusing on these bacteria could disrupt
their oncogenic roles across cancer types.
Implications of Induced Native Phage Therapy for
Oncology: A Microbiome-Centered Hypothesis
An expanding body of evidence indicates that many solid tumors harbor distinct, metabolically
active microbial communities within the tumor microenvironment (TME). These tumor-
associated bacteria are increasingly recognized as contributors to oncogenesis and tumor
persistence through mechanisms that include chronic inflammation, immune evasion, altered
antigen presentation, and metabolic reprogramming. Notably, enrichment of specific bacterial
taxa—such as Fusobacterium nucleatum—has been documented across multiple malignancies,
including colorectal, pancreatic, breast, gastric, and lung cancers, with mechanistic links to
immune suppression and therapy resistance.
In colorectal cancer, F. nucleatum has been shown to promote tumor progression through
adhesin-mediated interactions (e.g., FadA) that activate β-catenin signaling, as well as immune
evasion via Fap2-mediated engagement of inhibitory receptors on natural killer and T cells.
Similar microbial signatures have been identified in other tumor types, suggesting that bacterial
participation in the TME may represent a conserved, though context-dependent, oncogenic
cofactor rather than an isolated phenomenon.
These observations motivate investigation of therapeutic strategies that selectively disrupt tumor-
associated bacteria without broadly damaging host tissues or the commensal microbiome.
Bacteriophages are uniquely suited for this role, as they exhibit strain-restricted host specificity
and can replicate at sites where susceptible bacteria are present. Preclinical and translational
studies have demonstrated that phage-mediated targeting of tumor-associated bacteria can alter
immune infiltration, reduce pro-tumorigenic inflammation, and enhance responsiveness to
conventional therapies in experimental systems.
Induced Native Phage Therapy (INPT) extends this conceptual framework by proposing a non-
pharmacological method intended to recruit bacteriophages already present within the host
phageome, rather than administering exogenous phage preparations. Within this model, strain-
targeted electromagnetic signature sets are selected and encoded via Biospectral Emission
Sequencing (BES) with the objective of selectively engaging microbial targets and facilitating
conditions permissive for phage-mediated killing. Importantly, this framework does not require
that all tumor-associated bacteria be eliminated, nor does it presume direct cytotoxicity toward
malignant cells; rather, it hypothesizes that selective microbial reduction may indirectly remodel
the TME by attenuating inflammatory signaling, disrupting biofilm-like microbial niches, and
restoring immune surveillance as represented in Figure 1.
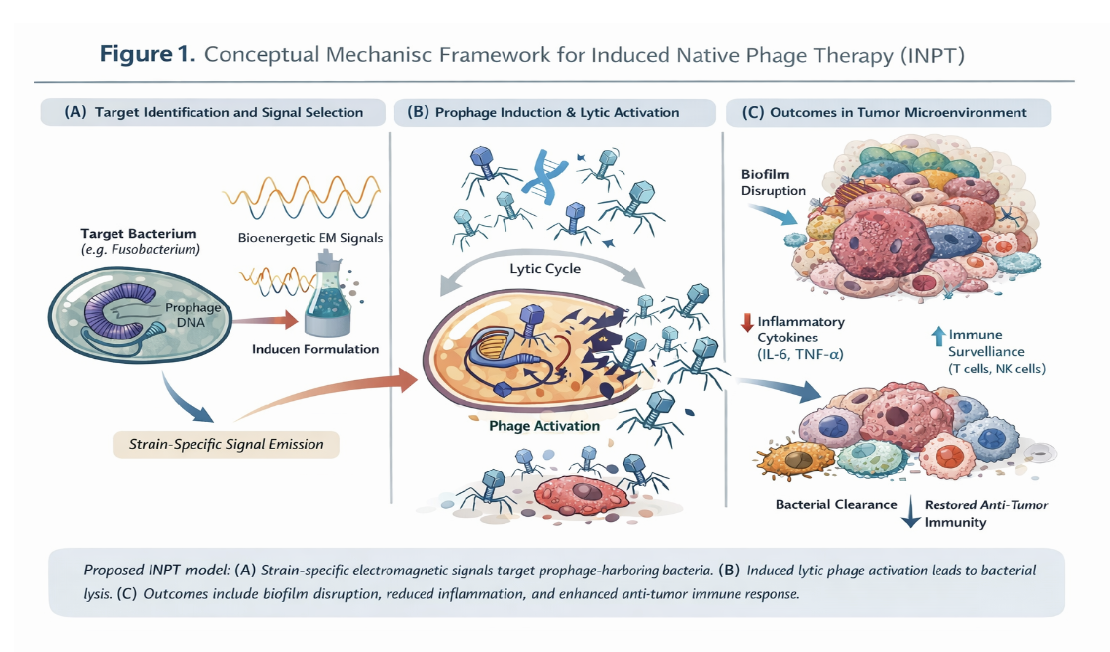
Figure 1 illustrates the proposed, hypothesis-consistent mechanism by which Induced Native
Phage Therapy (INPT) may selectively reduce pathogenic or tumor-associated bacterial
populations through recruitment of endogenous bacteriophages.
At present, the relevance of INPT to oncology remains a hypothesis grounded in biological
plausibility rather than demonstrated clinical efficacy inside of IRB-supported research
protocols. While infection-focused studies suggest rapid, strain-selective microbial clearance
consistent with phage-mediated dynamics, direct evidence of prophage induction, lytic transition,
and phage amplification within tumor-associated bacteria has not yet been established, due to the
nature of limited imaging capabilities of in vivo phage/microbe interactions.. Accordingly,
oncology applications of INPT is being evaluated through staged investigation, including tumor-
resolved microbiome and phageome profiling, assessment of prophage induction markers, spatial
immune-microbial mapping, and controlled clinical studies with predefined oncologic endpoints.
Nevertheless, the convergence of three independently established observations—(i) the detection
of the presence of tumor-associated bacteria across multiple cancers, (ii) the capacity of
bacteriophages to be induced through INPT to selectively eliminate specifically targeted
bacterial populations and amplify in situ, and (iii) the sensitivity of tumor immune dynamics to
microbial cues—supports the exploration of INPT as a microbiome-modulating adjunct rather
than a stand-alone anticancer therapy. If validated, such an approach could complement existing
modalities by addressing microbial drivers of immune suppression and therapeutic resistance that
are not directly targeted by the conventional cytotoxic or immunologic agents of oncology.


